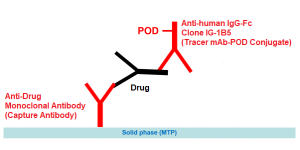
Enzyme immunoassay for the specific and quantitative determination of free Tocilizumab (Actemra®) in serum and plasma.
The solid phase (MTP) is coated by a highly specific monoclonal antibody directed against Tocilizumab. Another POD-labelled specific monoclonal antibody is used as conjugate.
Therefore any cross reactivity to the other therapeutical monoclonal antibodies is excluded.
Assay Format: Double specific mAb-based
Reference: IG-AB108
Barcode: 8682754930233
Legal Status: CE-Mark
Intended Use: This kit has been developed for the measurement of drug levels in research and diagnostic uses. It is suitable for Therapeutical Drug Monitoring (TDM) purposes.